Foot Implants: PEEK Devices for Foot & Ankle Applications
A Non-Metal Alternative for Extremity Applications
A metal-free solution for extremity applications

Increasing Adoption of PEEK-based Devices for Hand and Foot Applications
Delivering solutions for hand, foot & ankle applications

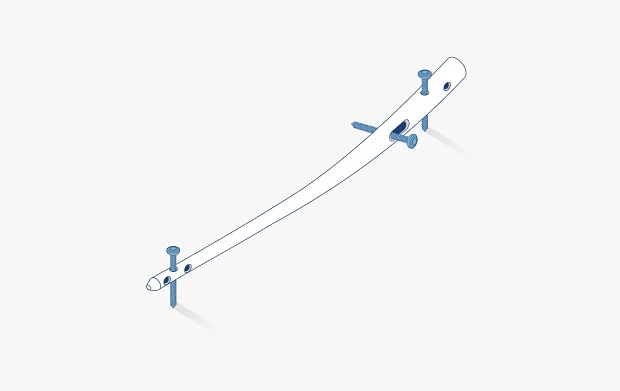
Pre-contoured anatomic plates
Both unfilled PEEK and carbon fiber reinforced PEEK materials have been successfully used in plating applications for the hand and foot.
Demonstrated benefits include:
- Bioinert and biocompatible with modulus of elasticity close to cortical bone
- Radiolucency allowing for a more accurate assessment of fusion1
- Improved patient satisfaction2

Hammertoe
PEEK-based intramedullary devices can provide similar advantages as metallic designs with potential additional benefits:
- the radiolucency of the material can enable an easier assessment of fusion
- the modulus of elasticity is very similar to bone, so there is minimal stress shielding at the arthrodesis site reducing risk factors that could disrupt normal bone healing5
- PEEK-based devices can simplify removal because it can be easier to saw than metals
- potential advantage in maintenance of correction because there is no risk of resorption of the graft material8
- Improved level of direct bone contact in as early as 4 weeks3
- New bone formation as early as 6 weeks4
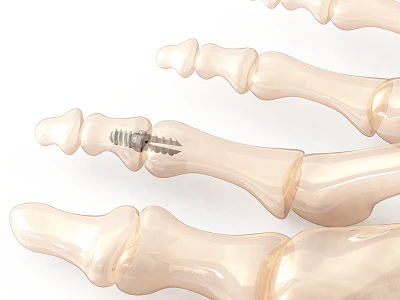
Image: Vector™ Hammertoe Correction System manufactured from PEEK-OPTIMA™ HA Enhanced, courtesy of Nvision Biomedical Technologies, Inc.™

Wedge osteotomy devices
Studies on implanted PEEK spacers for osteotomies in the foot demonstrate high fusion rates and conclude that PEEK offers:
- an effective alternative to structural bone grafts6,7
- potential advantage in maintenance of correction because there is no risk of resorption of the graft material8
- spacers can be designed as wedges to be used with ancillary fixation or as stand-alone devices with integrated screws
- Improved level of direct bone contact in as early as 4 weeks3
- New bone formation as early as 6 weeks4
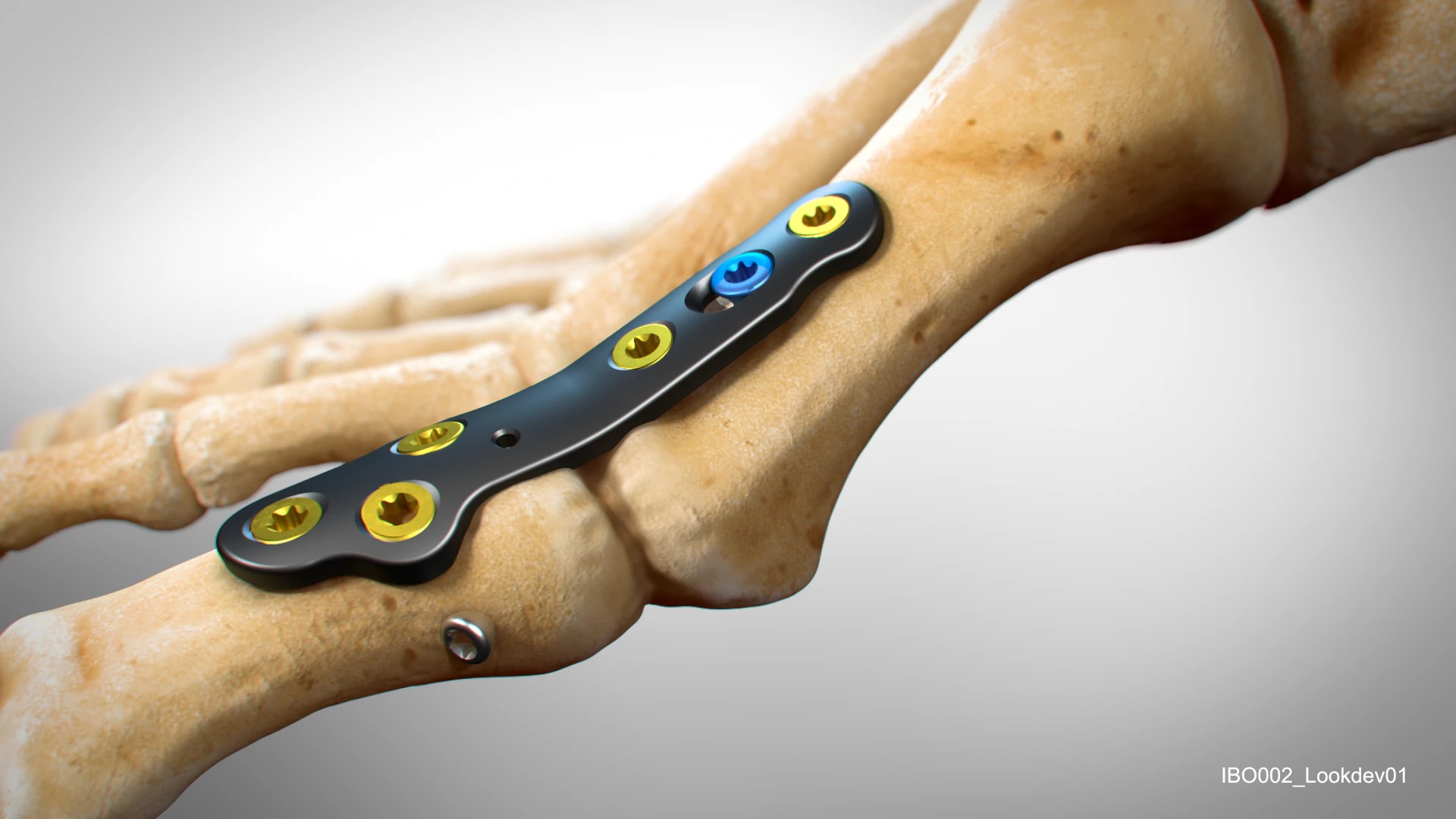
Image: The Trigon® Stand–Alone Wedge Fixation System, courtesy of Nvision Biomedical Technologies, Inc.™ Read more about it here
Pioneering composite polymer trauma plate solutions
By collaborating with leading trauma experts, innovators and implant manufacturers, we have developed a community who is passionate about advancing patient outcomes through high performance CFR-PEEK polymer solutions.
Featured resources

First FDA clearance for an osteotomy wedge system made of PEEK-OPTIMA™ HA Enhanced


References
1.Rhee PC, Shin AY. The rate of successful four-corner arthrodesis with a locking, dorsal circular polyether-ether-ketone (PEEK-Optima) plate. J Hand Surg Eur. Vol. 2013 Sep;38(7):767-73.
2. Harmer JL, et al. A Midterm Review of Lesser Toe Arthrodesis With an Intramedullary Implant. Foot Ankle Spec. 2017 Oct;10(5):458-464.
3. Walsh WR, et al. Does PEEK/HA Enhance Bone Formation Compared With PEEK in a Sheep Cervical Fusion Model? Clin Orthop Relat Res. 2016 Nov; 474(11): 2364–2372.
4. Study evaluated the in vivo response to PEEK-OPTIMA™ Natural, PEEK-OPTIMA™ HA Enhanced and allograft in a cervical spine fusion model in sheep. Data on file at Invibio. This data has not been correlated with human clinical experience.
5. Cook JJ, Cook EA. (2019). Proximal Interphalangeal Joint (PIPJ) Arthrodesis with a Poly-etheretherketone (PEEK) Implant. In: Cook E, Cook J (eds) Hammertoes.
Springer, Cham. https://doi.org/10.1007/978-3-319-16552-3_11
6. Gomez DN, Eslava S, Federico A, et al. Use of Poly(Ether Ether Ketone) cages in foot and ankle surgery. Foot Ankle Clin. 2012 Sep; 17(3): 449-57.
7. Zaghloul KM, Saied AM, Abouelnas BA, et al. Can polyaryletherketone cage be used to achieve union and maintain correction in anterior calcaneal lengthening
osteotomy for treatment of flexible flatfoot? J Pediatr Orthop B. 2019 Nov;28(6):598-601.
8. Pearson KT. Clinical Case Highlight: Tarsa-Link™ Stand-Alone Wedge Fixation System for flatfoot deformity repair – Evans osteotomy. http://www.
centricmedical.com/tarsalink-case-study-1. Accessed May 28, 2020.
9. 1. https://www.nextremity.com/new-research-indicates-up-to-60-million-americans-suffer-from-hammertoe-foot-deformities/. Accessed May 26, 2020
Trigon® Stand–Alone Wedge Fixation System for Cotton Osteotomies, image courtesy of Nvision Biomedical Technologies, Inc.™. Trigon® is a registered trademark of Nvision Biomedical Technologies, Inc.™
Vector™ Hammertoe Correction System manufactured from PEEK-OPTIMA™ HA Enhanced image courtesy of Nvision Biomedical Technologies, Inc.™ Vector® is a registered trademark of Nvision Biomedical Technologies, Inc.™