Active Implants & Drug Delivery
Accelerating new medical device design

Discover how to push the limits of drug delivery to the lungs in a concept for a next generation vaporizing inhaler made with PEEK

PEEK has the potential to drive innovation in smart, connected wearable health tech devices, for example in wearable patch pumps for diabetes management
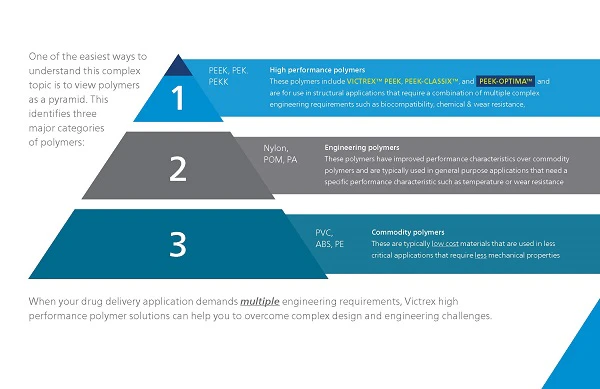
Trends driving innovation in medical devices
Drug delivery & active implantable medical devices have the potential to lower treatment costs, improve patient outcomes and promote an active lifestyle. Finding ways to improve the device or to develop new solutions is challenging – but with the right high performing materials, that journey can be easier. We are here to help you discover what is possible, and how far you can push the boundaries when inventing with PEEK.
Patient comfort
Manufacturers seek new technologies to help support patients with compliance, adherence and increase patient comfort
Wireless technology
Many new AIMD systems communicate wirelessly with the implant and other external equipment, and need to be minimally invasive
Smaller size batteries
Designers want to optimize the performance of ever increasingly miniaturized applications - how can devices be thinner and smaller whilst still retain a long battery life?
DEVICE APPLICATIONS
Neuromodulation
The dielectric properties of PEEK-OPTIMA polymer allow for exceptional performance as an insulator, with minimal interference and minimal loss over changing conditions such as temperature, humidity, and frequency.
Cochlear implants
Ventricular assist devices
PEEK-OPTIMA polymers have proven biocompatibility and resistance to a broad range of chemicals and blood, along with excellent wear resistance - and can be used to develop state-of-the art VAD such as total artificial hearts.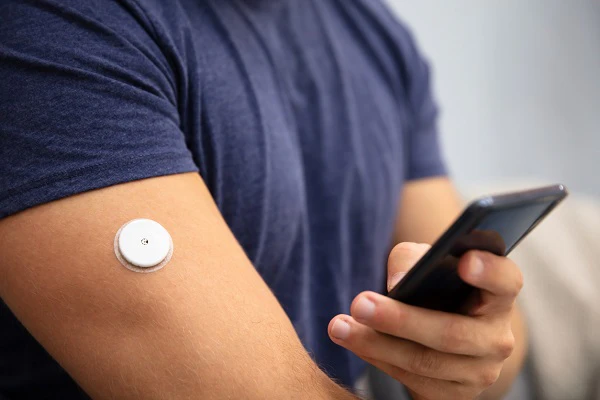
Drug delivery
Medical-grade PEEK polymers are biocompatible, are safe in contact with a range of drugs, chemicals and human tissue - and have the mechanical properties needed to innovate in devices such as auto-injector insulin pens, transdermal patch pumps, inhalers and more.
Need help with material selection?
Resources




1. Webinar: PEEK-OPTIMA™ HA Enhanced Devices for Cervical and Lumbar Fusion: Insights from Early Clinical Experience, presented by Timothy Bassett, MD and Brad Prybis, MD, March 2, 2017.
2. Study evaluated the bone ongrowth of PEEK-OPTIMA™ Natural and PEEK-OPTIMA™ HA Enhanced in a bone defect model in sheep. Data on file at Invibio. This has not been correlated with human clinical data.
3. Vadapalli S, Sairyo K, Goel VK, Robon M, Biyani A, Khandha A, Ebraheim NA. Biomechanical rationale for using polyetheretherketone (PEEK) spacers for lumbar interbody fusion-A finite element study. Spine 2006 Dec 15;31(26):E992-8.
4. Ziao Z, Wang L, Gong H, Zhu D. Biomechanical evaluation of three surgical scenarios of posterior lumbar interbody fusion by finite element analysis. Biomed Eng Online. 2012 Jun 18;11(1):31.