Transforming trauma with polymer composites
A leading innovator in trauma solutions

Learn about the advantages of partnering with Invibio as we support you from concept to commercialization in your next trauma device innovation.

Our carbon fiber composite trauma solutions are benefiting patients worldwide with a less stiff material compared to metal.

Carbon fiber composites vs. Stainless Steel
Imaging benefits across trauma patient care
Read about the intraoperative and postoperative benefits of radiolucency, and artifact-free imaging with implants manufactured with PEEK-OPTIMA Ultra-Reinforced
Setting the standard for over 20 years
Today, approximately 15 million PEEK-OPTIMATM devices are implanted worldwide.
Challenging patient health risk factors
Increasing global rises in high risk conditions that reduce the ability to heal after a fracture. Studies show that there is an elevated non-union risk associated with severe fractures in people with a high body mass index, and a history of smoking, and alcoholism.5
The rising cost of complications
Adverse events are 3 times more likely in trauma patients than other orthopedic patients, which is leading to more frequent and higher costs to revise and deal with complications.6
Reducing risk through device design
Medical device companies are seeking a new generation of high performance fixation devices that can offer adequate fatigue strength to withstand loads through the healing process, especially in patients with risk factors for implant related complications.
TRAUMA APPLICATIONS
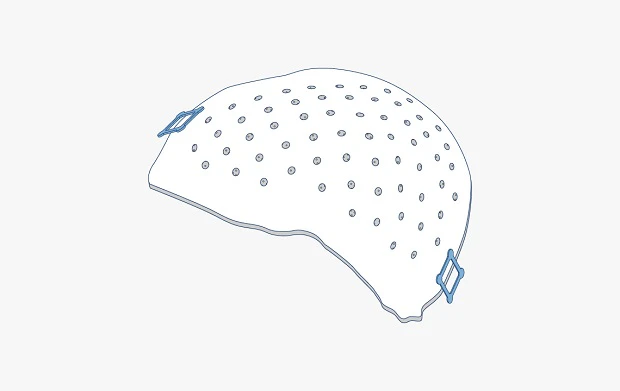
Patient-specific craniomaxillofacial implants
PEEK-OPTIMA high performance polymer is a strong, permanent, functional alternative to traditional structural materials like metal that can be used to manufacture custom cranial implants. PEEK-OPTIMA’s biomechanical properties are similar to bone, result in better clinical outcomes and have improved cosmetic satisfaction compared to titanium.

Sternal closure
Cerclage wires have been the standard of care for sternotomies but can result in loosening and patient discomfort when coughing. Poor bone quality in geriatric patients can also result in wire cut through and instability post operatively. Studies have shown rigid fixation results in better outcomes compared to standard cerclage wires. PEEK-OPTIMA polymers have been used to innovate in this space, with OEMs developing new metal-free technologies for fixation plate, ties and zips.

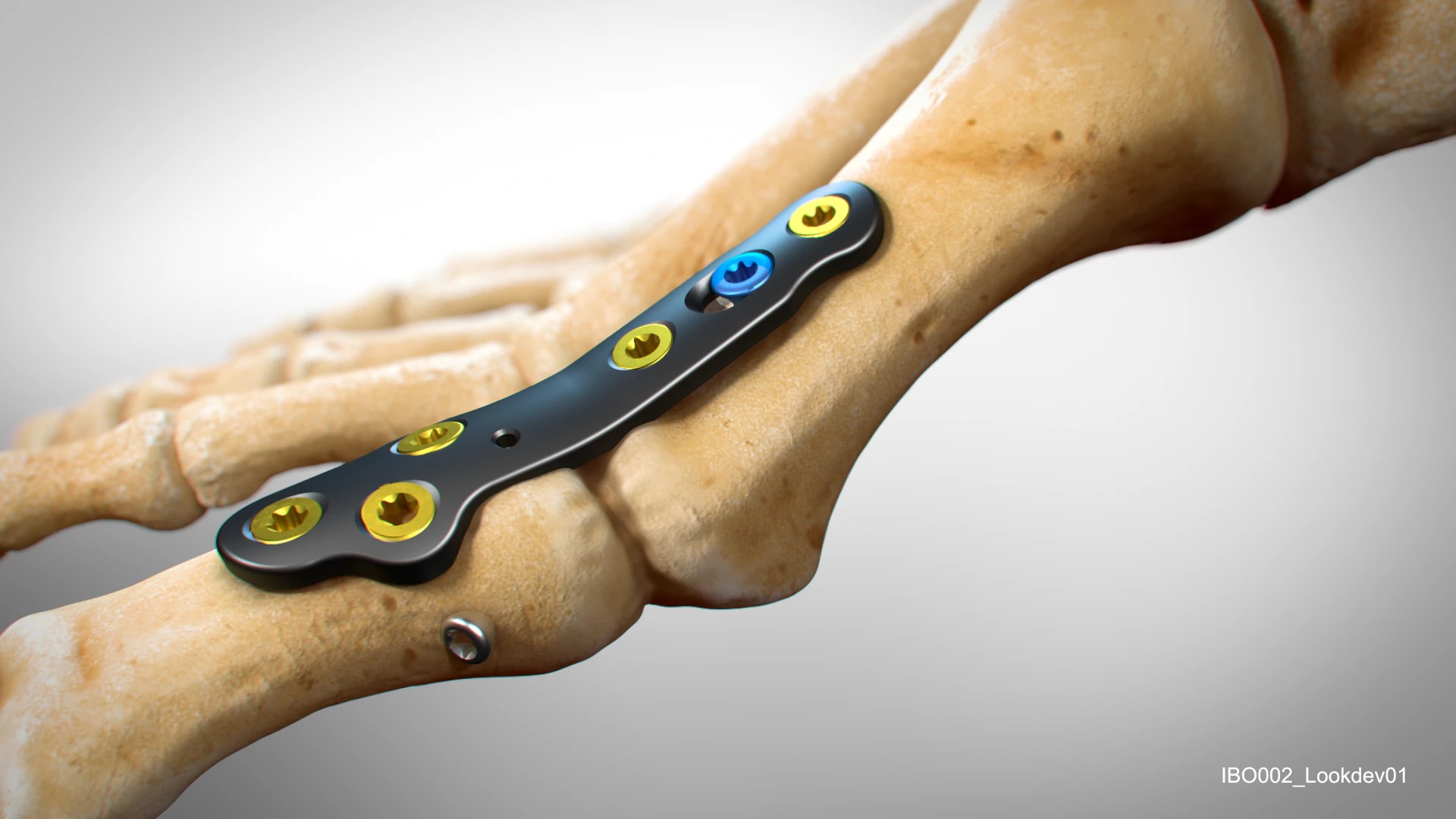
Fracture fixation plates
PEEK-OPTIMA Ultra-Reinforced combines the high-performance material properties of PEEK-OPTIMA Natural polymer with the strength imparted by continuous carbon fibers.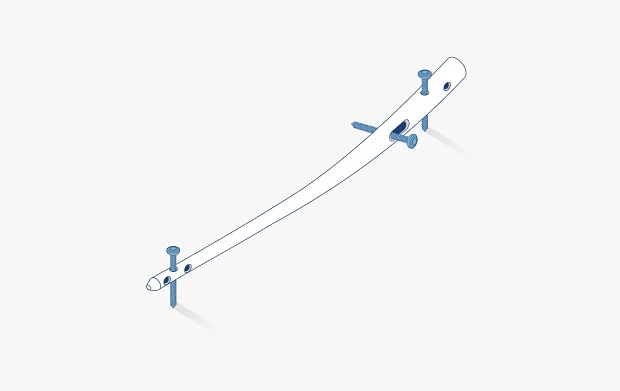
Intermedullary nails
PEEK-OPTIMA Ultra-Reinforced enables semi-rigid fixation with the potential for improved fatigue and imaging properties over metal2-4. In intermedullary nails, there is a greater percent of femoral nail cases healed at 6-week time period compared to reported rates of metal, and pain-free ambulation without assistive devices in 4-7 weeks in patients with tibia and fibula nails2-4Evidence in Trauma solutions





MEDICAL-GRADE MATERIALS
- Natural
- HA Enhanced
- Image Contrast
- Reinforced
- Ultra-Reinforced
- Natural
- HA Enhanced
- Image Contrast
- Reinforced
- Ultra-Reinforced
All the benefits of PEEK-OPTIMATM Natural plus Hydroxyapatite (HA) crystals embedded throughout the material, a well-known osteo-conductive material. Unlike interbody fusion devices that are coated, PEEK-OPTIMATM HA Enhanced devices have hydroxyapatite crystals on all surfaces which results in earlier bone on-growth and greater new bone formation on a fully osteoconductive implant. Key benefits include:
- Enhanced bone apposition1,
- Greater new bone formation2
- Higher quality of new bone bridging2.
- Natural
- HA Enhanced
- Image Contrast
- Reinforced
- Ultra-Reinforced
- Natural
- HA Enhanced
- Image Contrast
- Reinforced
- Ultra-Reinforced
A carbon fiber reinforced PEEK polymer compound to increase strength while reducing stress shielding by more closely matching cortical bone stiffness. Short carbon fibers are dispersed within the PEEK-OPTIMATM Natural matrix enhancing mechanical and physical properties for more demanding load-bearing implants and applications. Benefits include3:
- Fiber-to-matrix bond strength provides higher strength than PEEK-OPTIMA Natural Polymers
- Bone-like stiffness can reduce stress shielding by more closely matching cortical bone stiffness
- Extensive history of use in FDA 510(k) cleared, long-term implantable devices
- Natural
- HA Enhanced
- Image Contrast
- Reinforced
- Ultra-Reinforced
An innovative composite formed by continuous carbon fibers dispersed within the PEEK-OPTIMA Natural polymer matrix to enhance mechanical strength and stiffness.
Benefits include:
- Excellent fatigue resistance and structural strength for device longevity4
- Tailored stiffness for design flexibility4
- Radiolucent, offering circumferential visibility of the surgical site before and after the procedure.
References for PEEK-OPTIMATM materials
1. Study evaluated the bone ongrowth of PEEK-OPTIMA Natural and PEEK-OPTIMA HA Enhanced in a bone defect model in sheep. Data on file at Invibio. This has not been correlated with human clinical data.
2. Study evaluated the in vivo response to PEEK-OPTIMA Natural, PEEK-OPTIMA HA Enhanced and allograft in a cervical spine fusion model in sheep. Data on file at Invibio. This data has not been correlated with human clinical experience.
3. Supporting information available upon request.
4. Data on file at Invibio Biomaterial Solutions™. Mechanical Benchmark of Carbon Fiber PEEK-OPTIMA™ Ultra-Reinforced vs Ti 6AI-4V Plates undergoing Static and Dynamic Testing per ASTM F382-99 (2008).
References
1.Data on file at Invibio Biomaterial Solutions. Mechanical Benchmark of Carbon Fiber PEEK-OPTIMA™ Ultra-Reinforced vs Ti 6Al-4V Plates undergoing Static and Dynamic Testing per ASTM F382-99 (2008).
2.Ziran BH, Mansour J, Jahangir AA, Min W. Efficacy of Carbon Fiber-Reinforced PEEK Femoral Nail Preliminary Results. Poster #68. Presented at OTA 2017.
3.Allison DC, Menendez LR. Carbon fiber fixation in oncologic bone surgery. Presented at MSTS Meeting 2013. Carbon Fiber Intramedullary Nail Fixation in Troublesome Long Bone Fractures and Nonunions in Cedars-Sinai Advances in Orthopaedics Newsletter, Spring 2014.
4.Zimel MN, Hwang S, Riedel ER Healy JH. Carbon fiber intramedullary nails reduce artifact in postoperative advanced imaging. Skeletal Radiol. 2015 Sep;44(9):1317-25.
5.Zura R, Xiong Z, Einhorn T, Watson JT, Ostrum RF, Prayson MJ, Della Rocca GJ, Mehta S, McKinley T, Wang Z, Steen RG. Epidemiology of Fracture Nonunion in 18 Human Bones. JAMA Surg. 2016 Nov 16;151(11):e162775. doi: 10.1001/jamasurg.2016.2775. Epub 2016 Nov 16. PMID: 27603155.
6.Sathiyakumar V, Thakore RV, Greenberg SE, Whiting PS, Molina CS, Obremskey WT, Sethi MK. Adverse Events in Orthopaedics: Is Trauma More Risky? An Analysis of the NSQIP Data. J Orthop Trauma. 2015 Jul;29(7):337-41. doi: 10.1097/BOT.0000000000000293. PMID: 26091531.