Common questions about PEEK for medical implants
How does PEEK compare to titanium for spinal cages?
What makes PEEK suitable for permanent implants?
What regulatory clearances have PEEK-OPTIMA been used in?
Medical Applications of Invibio PEEK solutions

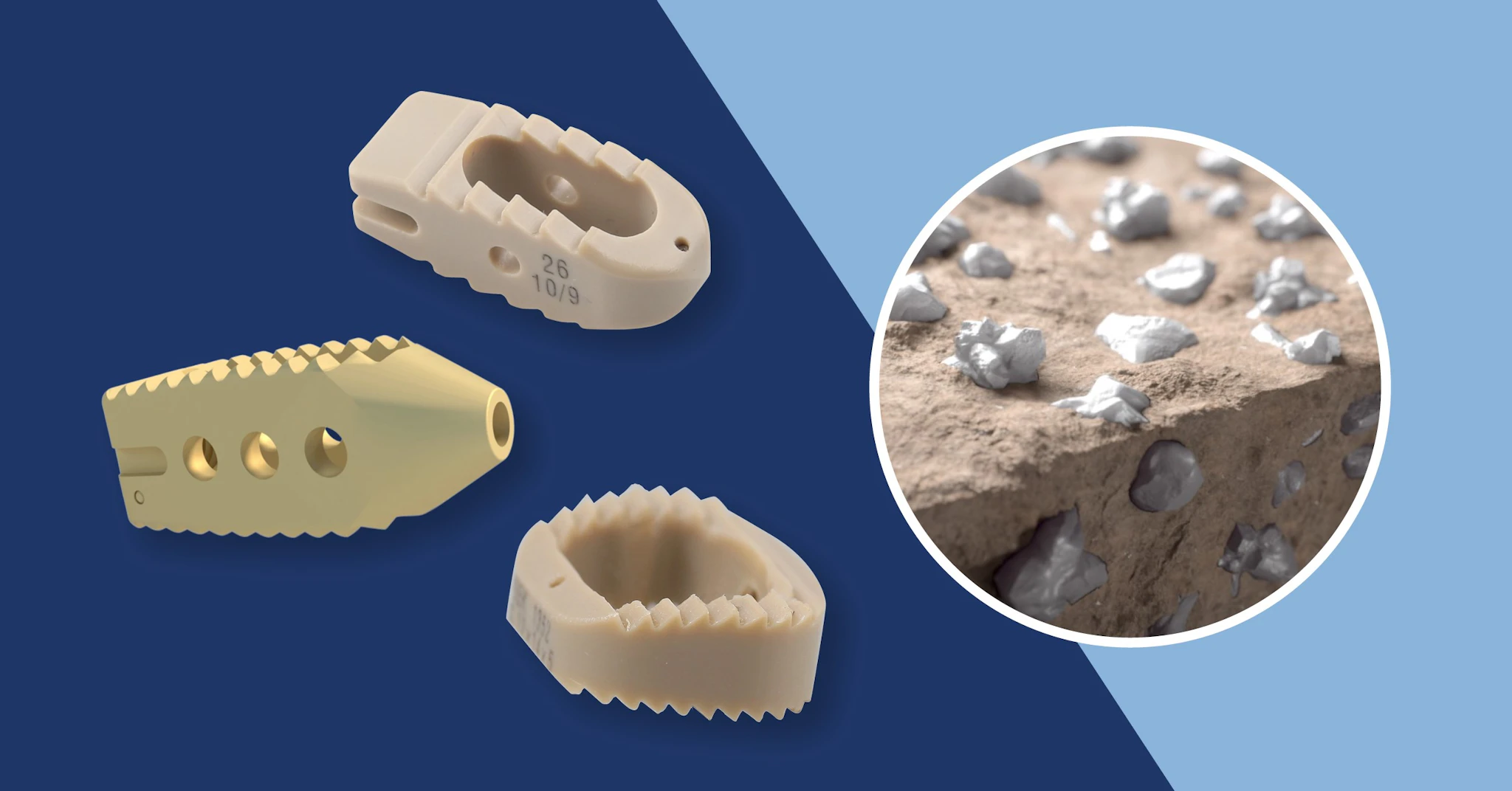
Spine & Orthopaedics
Invibio provides PEEK-OPTIMA™ polymers for implants that restore mobility and support bone integration in spinal and joint procedures. These materials combine mechanical strength with radiolucency for precise surgical assessment.
Applications:
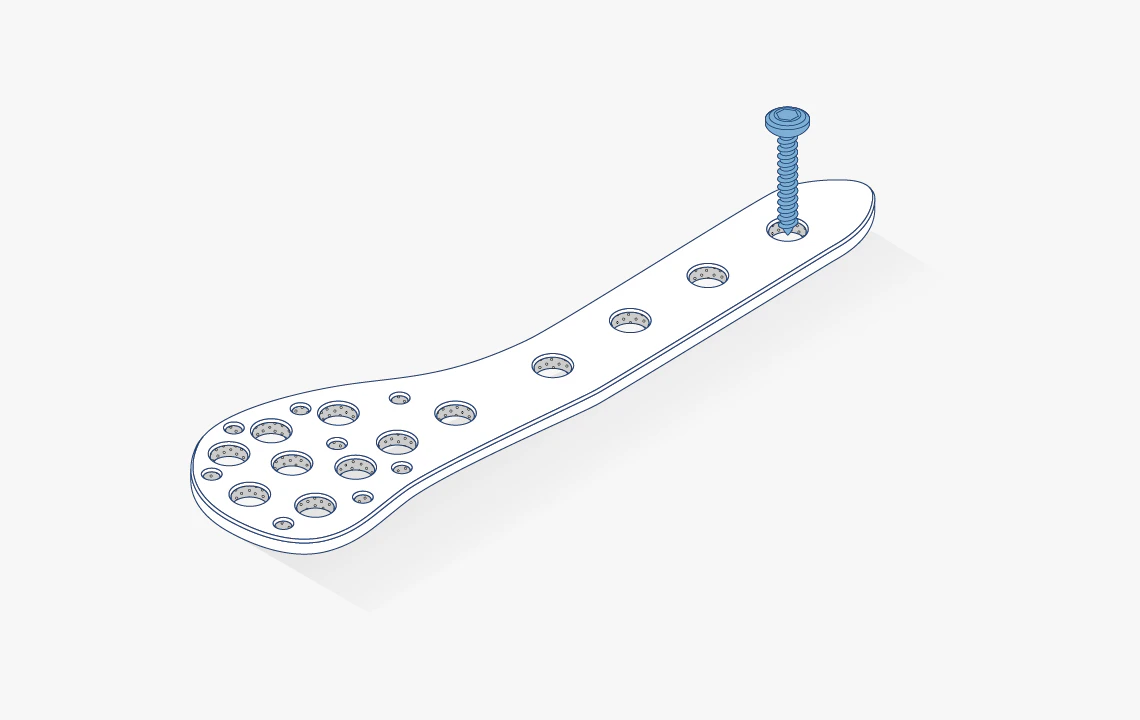
Trauma
Our PEEK-based composites and reinforced materials are developed for trauma repair and reconstruction. They enable lighter, more flexible devices that maintain high strength and imaging compatibility.
Applications:
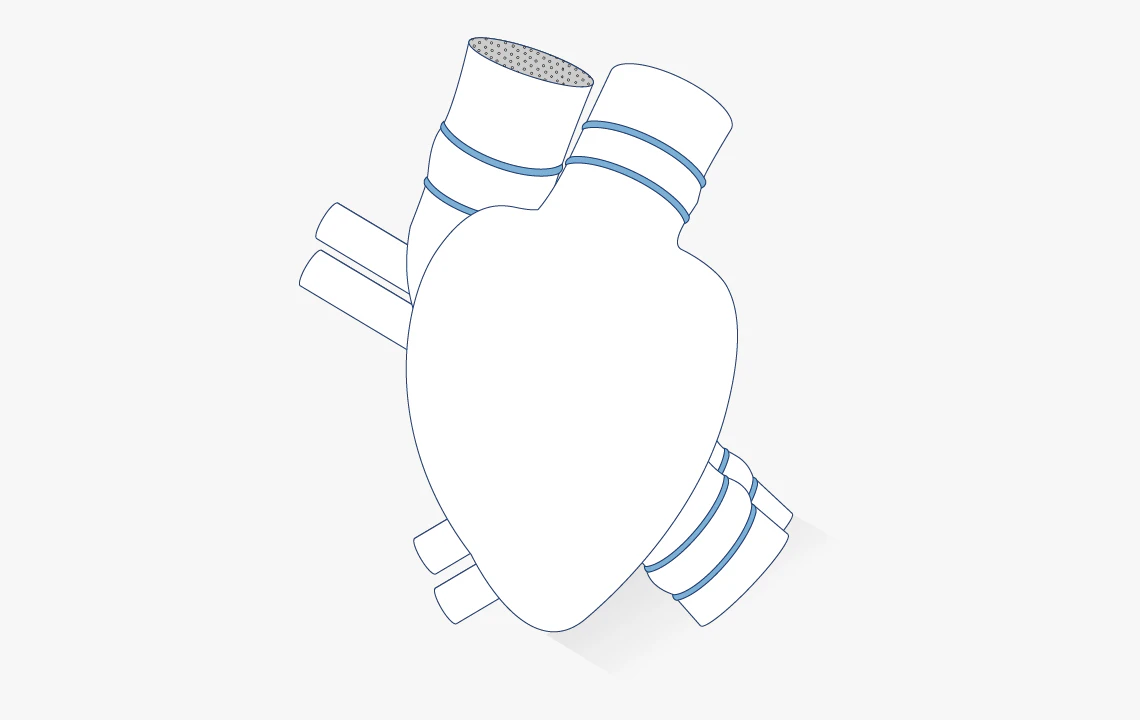
Medical devices
Invibio supports medical device manufacturers with implant-grade polymers for advanced applications such as controlled drug delivery and active implant systems.
Applications:

Creating Value in Medical
Invibio was a pioneer as the very first company to make PEEK polymers available for medical devices. Our goal is to find areas where our products deliver a clinical benefit that’s better for the patient, or an economic benefit where our solutions can reduce healthcare costs around the globe.