Remember when titanium was the best CMF material you could offer your patients?
Implant Options
The key clinical challenges in reconstruction of craniofacial bone defects today are:
- Infection
- Implant fit
- Ability to carry out complex reconstructions
- Aesthetic appearance
There is a growing surgeon awareness to overcome these clinical challenges by using prefabricated custom implants for cranioplasty procedures. Today’s custom implants employ the latest advances in imaging, coupled with 3D modelling and state of the art manufacturing to produce the final implant with perfect fit. A number of material options are at the disposal of the surgeon when it comes to cranioplasty. Among these are:
3 Clinical Outcomes with Custom PEEK Implants
In comparison with autologous bone, custom PEEK implants have been shown to result in significantly lower complication rates8 and, in a study of PEEK-OPTIMA Natural vs. titanium mesh (+/- acrylic cement), Ng et al. found no complications in the PEEK group, but 80% and 43% in the titanium and titanium plus acrylic groups, respectively. Similarly, there were no failures in the PEEK group, but 60% and 43% in the titanium and titanium plus acrylic groups, respectively.2,5
1. Avoiding post-operative infection
Another large study of 132 patients receiving PEEK-OPTIMA Natural or titanium mesh implants, found that overall complication rates, including infection, were similar between the groups, but there was a trend towards increased exposure of the titanium implants.3 Although infection rates can be dependent upon a number of factors, including length of surgery and proximity to the frontal sinus, the rate associated with PEEK cranioplasty is similar to that of prefabricated PMMA and titanium plates, but lower than that observed for autologous bone.2,6
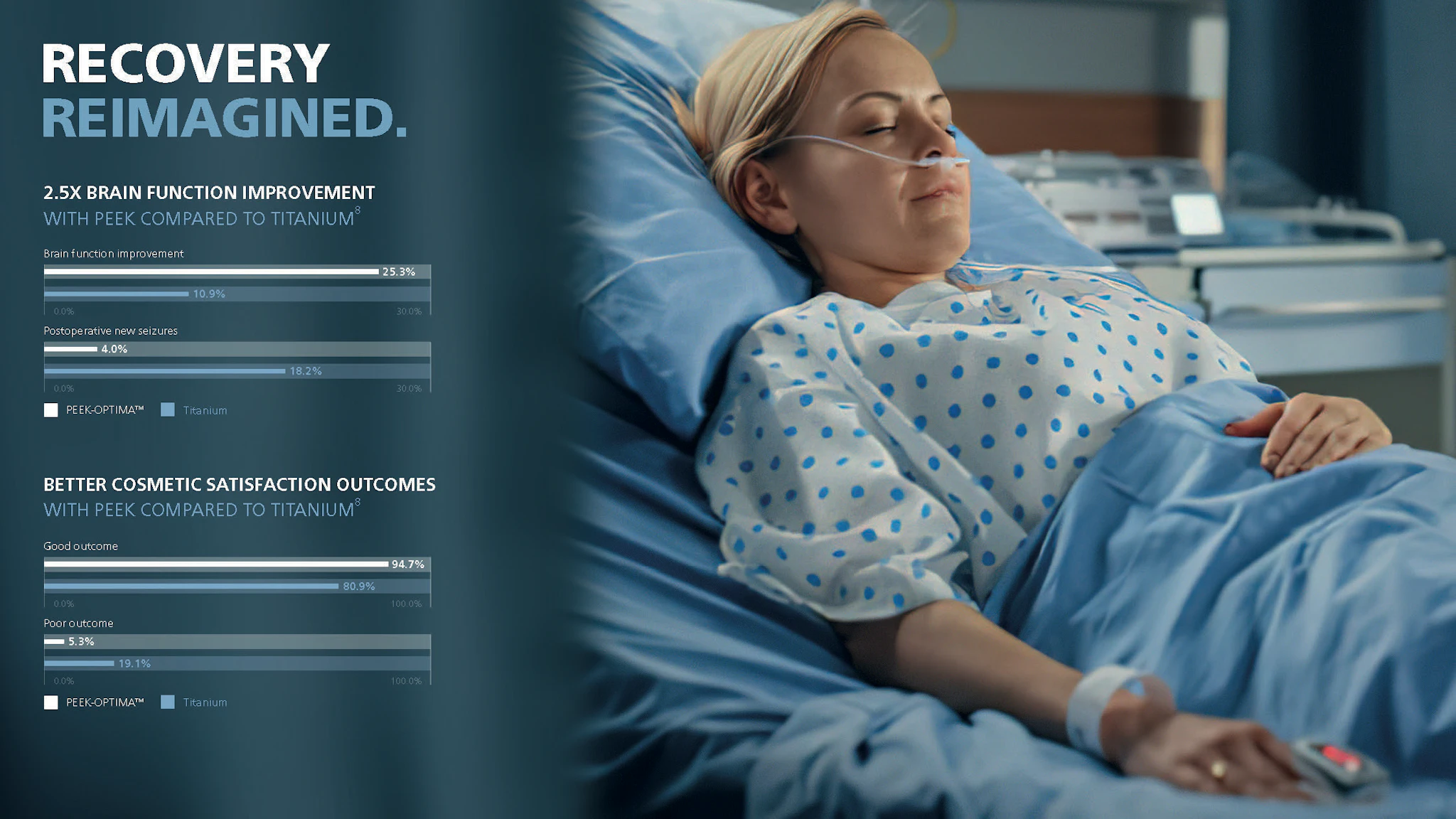
2. Improving brain function and reducing implant exposure
In probably the largest, multi-center retrospective study to date comparing PEEK-OPTIMA Natural with titanium, Zhang et al. followed a total of 185 patients.4 The indication for the majority of patients in each group was trauma (82.7% in the PEEK group vs. 70.0% in the titanium mesh group), and most involved complex reconstructions (78.7% in the PEEK group vs. 80.9% in the titanium group). The authors identified a number of significant differences between the groups. Among these, brain function improvement was higher in patients receiving a PEEK implant (25.3%) compared with titanium implant (10.9%), and the PEEK group displayed a significantly lower rate of complications overall (17.3%) compared with the titanium mesh group (31.8%). Of note also, was the higher incidence of post-operative implant exposure in the titanium group which can, in many cases, necessitate re-operation. A similar propensity for wound breakdown and implant exposure of titanium implants compared with PEEK has been seen in other studies.3,25
3. Reducing risk of complications
Finally, a recent systematic review and meta-analysis looked at the combined literature for PEEK cranioplasty and compared it with the outcomes of titanium mesh and autologous bone.27 In the meta-analysis comparing PEEK with autologous bone, there were no instances of complication in the PEEK group, while the complication rate in the bone group was 37%, representing a 7.69-fold increase in the odds of complication with autologous bone. Similarly, there was a 1.74-fold increase in the odds of implant failure in the autologous bone group compared with PEEK (10.9% vs. 0% respectively).
The second meta-analysis included in this study comparing with titanium mesh revealed similar advantages with PEEK, including a 7.87-fold increase in the odds of post-operative complication with titanium (38.1% vs. 16.7% for PEEK) and a 5.88-fold increase in the odds of implant failure with titanium (26.5% vs. 8.3% for PEEK). Implant infection was seen in 6% of PEEK cases, which was towards the lower or midpoint of the ranges cited by the authors in the literature (0-25.9% for bone and 0-11% for titanium mesh). Similarly, the overall complication rate (15.3%) and failure rate (8.7%) for PEEK implants were towards the lower end cited in the literature for autologous bone and titanium mesh.
The authors concluded that there was a trend towards lower post-operative complications with PEEK compared with autologous bone as well as a trend towards lower implant failure with PEEK compared with titanium.27 Although these trends did not quite reach statistical significance (probably due to the small number of studies and data available), this is the first study employing statistical analysis of multiple studies that points towards potentially advantageous clinical outcomes for PEEK cranioplasty patients.