Active implantable medical devices
Technology to stimulate innovation
Proven clinical history
Versatile polymers for a range of next-generation components
PEEK-OPTIMATM polymers have the potential to be used in a broad range of applications including IPG housings & enclosures, neural interfaces, lead and leadless devices and telemetry components
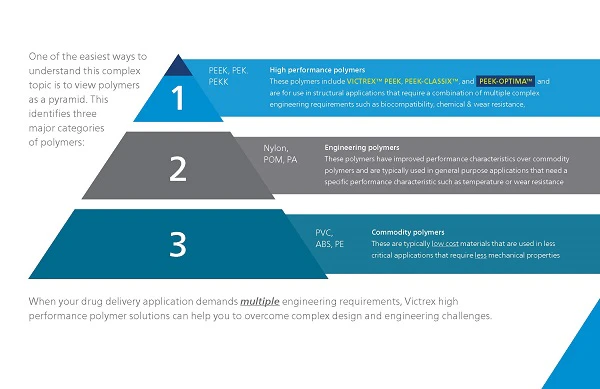
Material selection
Transformative PEEK implant material for medical applications
Biocompatibility
Masterfile of biocompatibility and clinical evidence, from over 15 million devices implanted over 20 years, in a wide range of human tissue types.
Minimal electrical interference & loss
Exceptional performance as an insulator, with minimal interference and minimal loss over changing conditions such as temperature, humidity, and frequency.
Electrical stability
Very stable dielectric properties (Dk, Df) over a wide range of temperatures, humidities and frequencies to support connectivity and telemetry
Improved visibility under imaging
Radiolucent in X-rays, MRI and other imaging
Hydrolysis resistance
Low moisture absorption (0.05wt%) means components retain dimensions in biological or fluid environments
Lightweight & durable
Able to resist impact, and for continued use over time, even in miniaturised parts under load

Article: PEEK polymer - a good choice for active implantable medical devices?
Whenever a challenging combination of material properties is required in implantable devices, high-performing PEEK polymers offer a range of compelling mechanical, chemical and electrical attributes & biocompatibility.
Discover more about PEEK solutions



