Benefits
Material
PEEK-OPTIMA™
Ultra-Reinforced Polymer
Resources
Featured resources
More applications

Dr Fedra Zaribaf, Senior Parts Engineer at Invibio explores the potential of carbon fiber PEEK composites in post-surgical bone healing.
PEEK-OPTIMA™ Ultra-Reinforced polymer offers a non-metal fracture fixation technology that provides strength and fatigue resistance demanded by high-load implant applications. Benefits include:
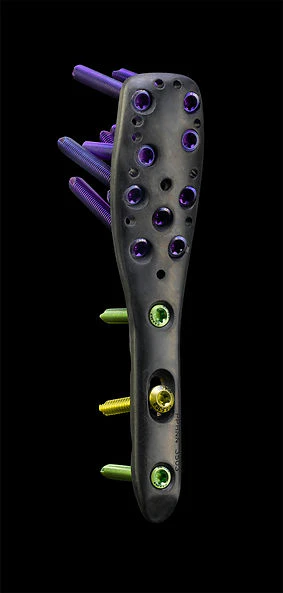
David J. Hak, MD, MBA, FACS: Effect of Implant Stiffness on Fracture Healing1

PEEK-based devices for hand and foot applications are experiencing an increase in adoption, with growing clinical evidence demonstrating the benefits PEEK can offer.
References
1. Image: Proximal Humeral Plate made with PEEK-OPTIMA polymers, courtesy of CarboFix Orthopedics Ltd
2. Data on file at Invibio. Mechanical Benchmark of Carbon Fiber PEEK-OPTIMA™ Ultra-Reinforced vs Ti 6AI-4V Plates undergoing Static and Dynamic Testing per ASTM F382-99 (2008).
3. Caforio M, Perugia D, Colombo M, Calori GM, Maniscalco P. Preliminary experience with Piccolo Composite™, a radiolucent distal fibula plate, in ankle fractures. Injury. 2014 Dec;45 Suppl 6:S36-S38.
4. Tarallo L, Mugnai R, Adani R, Zambianchi F, Catani F, A New Volar Plate made of Carbon-Fiber-Reinforced Polyetheretherketone for Distal Radius Fracture: Analysis of 40 Cases, J Orthop Traumatol, 2014 Dec; 15(4): 277-83