Megatrends
Setting the standard for over 20 years
Ageing global population
People are living longer and have a strong desire to maintain their quality of life in their later years.
Joint replacement & pain management
Extended life expectancy results in an increasing need to replace worn out body parts or to alleviate pain in order to resume normal activities.
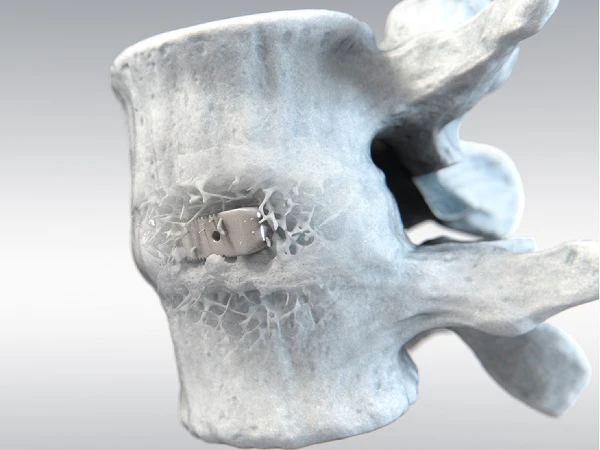
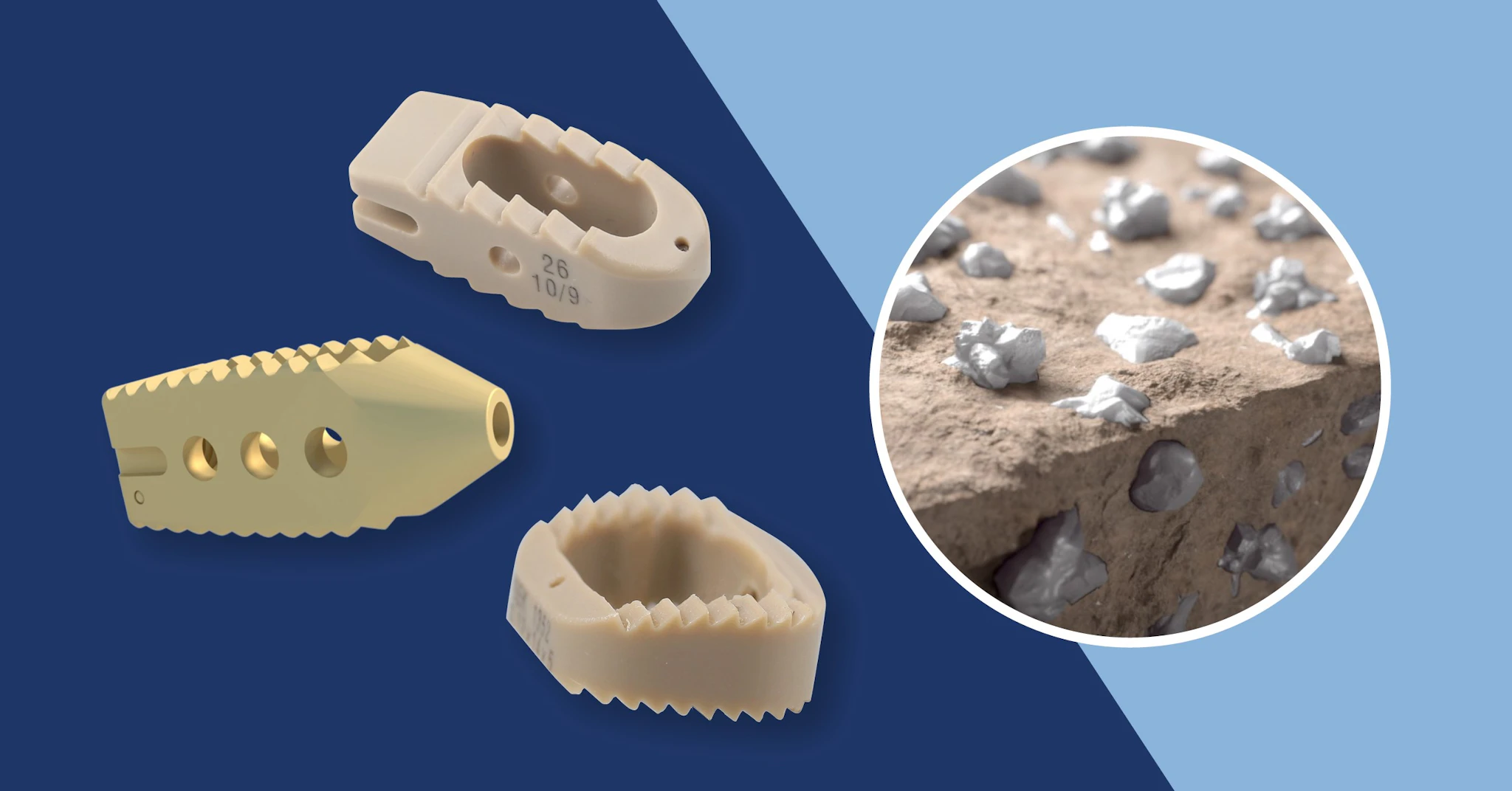
High performance solutions providing societal benefits
Invibio provides solutions that can be used in a minimally invasive manner, helping to enhance clinical benefits.
Interested in evaluating
PEEK-OPTIMATM polymers for your next device?
Resources
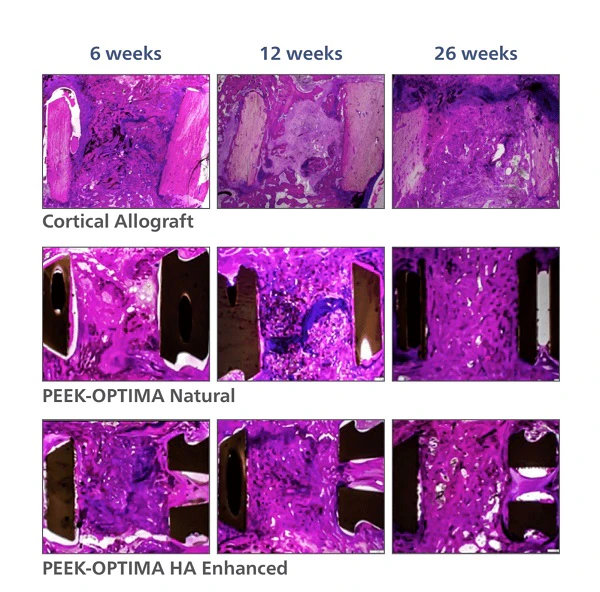
Evidence in Spine & Orthopedic solutions
References
1. IQVIA, Spinemarket Inc. 2018, Minneapolis, MN